The CRO Compliance & Governance Challenge: Complex Trials, High Stakes
CROs operate in one of the most tightly scrutinised environments in the world.
Between ICH-GCP, EU CTR, FDA/EMA/MHRA requirements, GVP, GDPR/HIPAA, 21 CFR Part 11, and sponsor quality agreements, it’s no longer enough to say you follow GCP — you must prove it, continuously.
Meanwhile, clinical portfolios are more global, decentralised, and data-heavy than ever.
Common pain points for CRO teams include:
⚠️ Fragmented quality, risk, and compliance data across studies, sponsors, and regions
⚠️ Manual tracking of audits, CAPAs, deviations, and inspections in spreadsheets
⚠️ Difficulty mapping controls to GCP, CTR, GVP, GDPR, ISO 27001, and sponsor requirements
⚠️ Limited visibility of operational and compliance risk across portfolios
⚠️ High cost and stress of sponsor audits and regulatory inspections
⚠️ Inconsistent documentation of SOPs, training, and trial documentation
⚠️ New decentralised/virtual models outpacing existing governance
One major inspection finding can jeopardise sponsor trust, patient safety, and future awards.
What Governance, Risk & Compliance Software Does for CROs
GRC software gives CROs a single system of record for SOPs, risks, obligations, and evidence — turning scattered QA and compliance activity into a structured, auditable framework.
With ISMS.online, CROs can:
✅ Centralise governance & SOPs — one hub for every policy, procedure, and control.
✅ Simplify inspection and audit readiness — prepare for sponsor, QA, and regulatory inspections from a single environment.
✅ Map controls to frameworks & sponsors — align to ICH-GCP, CTR, GVP, GDPR/HIPAA, ISO 27001, and sponsor quality agreements.
✅ Enhance risk visibility — dashboards show risk by study, programme, region, or function.
✅ Standardise quality management — keep governance consistent as portfolios and service lines grow.
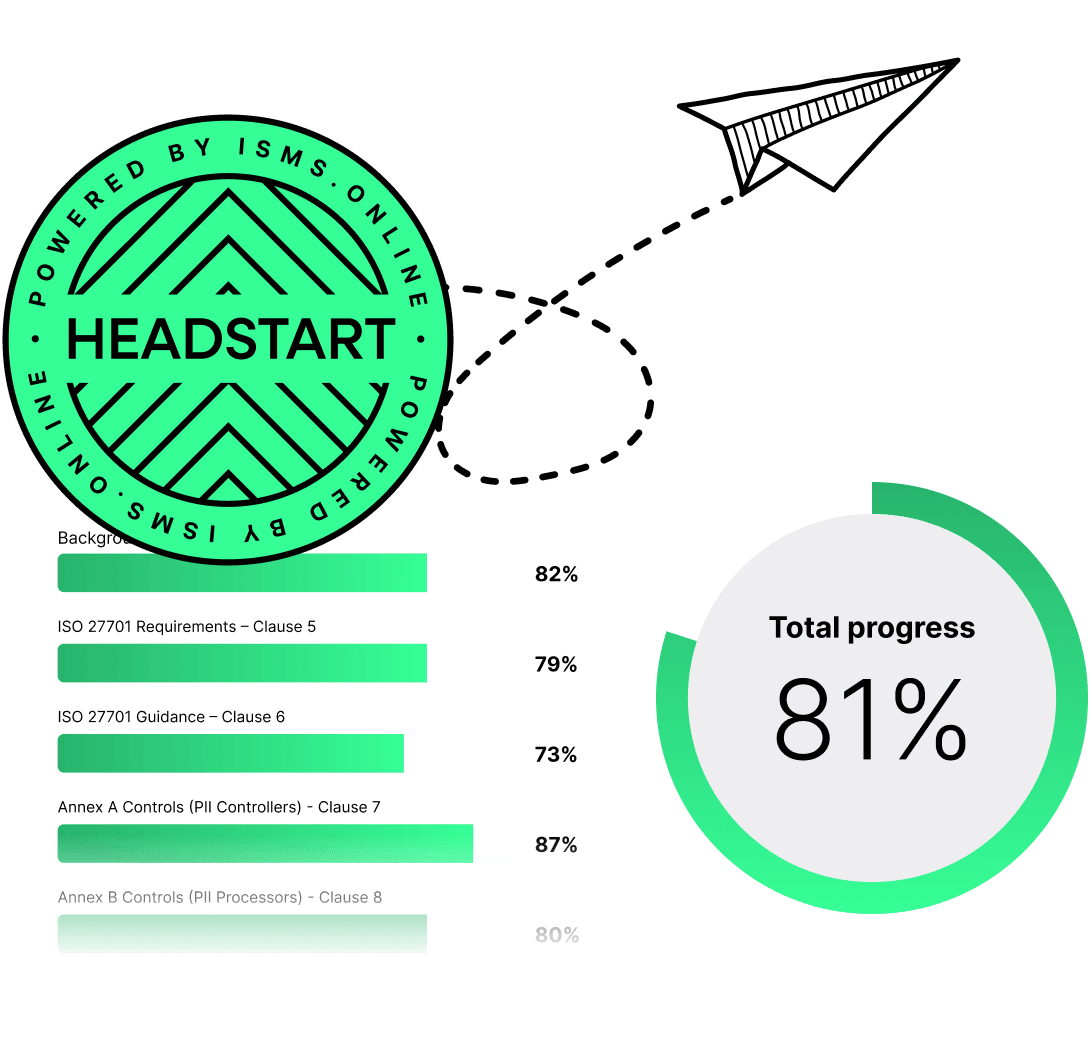
ISO 27001 made easy
An 81% Headstart from day one
We’ve done the hard work for you, giving you an 81% Headstart from the moment you log on. All you have to do is fill in the blanks.

Meet ISMS.online — The All-in-One GRC Platform Built for CROs
ISMS.online empowers CROs to manage quality, risk, and compliance with confidence — without drowning QA and operations teams in manual admin.
Purpose-built for global clinical research organisations:
🧩 Pre-mapped to key frameworks (ICH-GCP, EU CTR, GVP, GDPR/UK GDPR, HIPAA, ISO 27001, ISO 9001, 21 CFR Part 11)
⚙️ Configurable workflows for audits, CAPAs, deviations, approvals, and sign-offs
🔗 Integrates with CTMS, EDC, eTMF, ticketing, and identity systems
📁 Evidence repository with full audit trails, version control, and inspection traceability
📊 Real-time dashboards for quality metrics, risk posture, and compliance progress
🌍 Supports multi-study, multi-sponsor, and multi-region operations in a single platform
From Pain to Process: Turn Compliance Burdens into Sponsor Confidence
You’re tracking audits, CAPAs, and deviations in disconnected spreadsheets.
→ ISMS.online centralises quality events, actions, and evidence in one system.
Result: faster, cleaner inspections and reduced inspection findings.
You struggle to prove consistent control to sponsors and regulators.
→ Evidence, SOPs, and audit trails are mapped to specific requirements.
Result: stronger sponsor trust and fewer surprises during audits.
You lack a clear view of risk by study, sponsor, or region.
→ Dashboards highlight risk and compliance status across programmes and functions.
Result: better decisions, prioritised CAPAs, and proactive oversight.
You’re rolling out decentralised or virtual trial models.
→ Standardised frameworks and workflows keep governance aligned with new delivery models.
Result: safer innovation and less regulatory friction.
How CRO Teams Use ISMS.online
Preparing for Regulatory Inspections & Sponsor Audits
Consolidate SOPs, risk registers, audits, and CAPAs in one place.
✅ Reduce inspection prep time and respond confidently to findings.
Managing CAPAs, Deviations & Quality Events Across Studies
Log, assign, and track all quality events to closure.
✅ Demonstrate effective root cause analysis and continual improvement.
Overseeing Data Protection, BAAs & Cross-Border Data Flows
Manage GDPR/HIPAA controls, BAAs, and data transfer mechanisms.
✅ Protect subject data and meet sponsor and regulator expectations.
Tracking Training, SOP Adherence & Role-Based Responsibilities
Link training records and responsibilities to SOPs and roles.
✅ Show that processes are not only defined, but followed.
Coordinating Risk Registers Across Programmes & Service Lines
Maintain structured risk assessments by study, function, or service.
✅ Build a portfolio view of operational and compliance risk.
Reporting to Sponsors, QA Leadership & Boards
Generate dashboards and reports for internal and external stakeholders.
✅ Provide transparent, up-to-date views of quality and compliance performance.

Free yourself from a mountain of spreadsheets
Embed, expand and scale your compliance, without the mess. IO gives you the resilience and confidence to grow securely.

Simple, Guided Onboarding — From Setup to Inspection-Ready
1️⃣ Discovery — Map studies, sponsors, functions, systems, and obligations.
2️⃣ Configure — Tailor templates and workflows to your models, regions, and sponsors.
3️⃣ Migrate — Import SOPs, risk registers, audits, CAPAs, and historical evidence.
4️⃣ Train — Enable QA, clinical operations, PV, data, and IT teams with guided support.
5️⃣ Optimise — Use dashboards and reports to drive oversight and continual improvement.
“You’ll be supported by real compliance experts — not bots — every step of the way.”
Flexible Plans for CRO Growth
Whether you’re a specialist CRO or a global full-service provider, ISMS.online scales with your portfolio.
Starter Plan — for specialist or early-stage CROs
- Fast-track to structured governance, risk, and quality management.
Growth Plan — for multi-sponsor, multi-region CROs
- Multi-entity, multi-framework governance with richer reporting.
Enterprise Plan — for global CROs with large study portfolios
- Advanced automation, complex obligation mapping, and enterprise integrations.
See ISMS.online in Action for CROs
Protect patients and data. Strengthen your governance.
Deliver the trust your sponsors, regulators, and partners expect. Learn how ISMS.online can help your organisation by booking a live demonstration.
FAQ: What CRO Teams Ask Before They Switch
How long does implementation take across multiple studies and regions?
Most CROs are live within 4–6 weeks, with full operational use typically under 8 weeks.
Can we manage GCP, CTR, GVP, GDPR, and ISO 27001 in one platform?
Yes — ISMS.online supports unified governance across quality, regulatory, privacy, and security frameworks.
Does it integrate with our CTMS, EDC, ticketing, and IAM tools?
Yes — integrations are available for major eClinical, service desk, and identity platforms.
Will sponsors, auditors, and regulators accept evidence from ISMS.online?
Yes — the platform is designed around recognised standards and is trusted by auditors and global sponsors.
How is data hosted and protected?
ISMS.online is hosted in ISO 27001-certified UK & EU data centres with strong encryption and full GDPR compliance.
Can we manage both traditional and decentralised trials?
Absolutely — you can model risks, controls, and obligations across traditional, hybrid, and decentralised study designs.
Can we support new service lines and complex partnerships safely?
Yes — reusable templates and workflows make it easy to extend governance to new offerings and collaborations.









