Why ISO 27001 Compliance Software is Critical for CROs
Clinical programmes race against milestones while sponsors and authorities tighten scrutiny. System sprawl (EDC, eTMF, CTMS, IRT/RTSM, LIMS, DCT) scatters proofs of control when an inspection or sponsor audit lands. Third-party dependencies broaden the blast radius if ownership is unclear. Audit sprints drain capacity and leave brittle systems that crack under the next CAPA.
- System sprawl fragments evidence and slows inspectors.
- Manual evidence hunts delay FDA/EMA/MHRA inspections and sponsor qualification.
- Undefined owners across functions cause remediation drift.
- CSV/Part 11 burden creates inconsistent validation and sign-off.
- Audit trail reviews are ad-hoc, exposing data-integrity risk.
- TMF completeness fluctuates, creating closeout friction.
- DCT vendors (eCOA/ePRO/eConsent) vary by study, making oversight brittle.
An ISO-first operating system resolves these pains by linking risks, controls, assets, owners and evidence into one narrative, making ownership visible and readiness continuous.
Regulatory Alignment with ISO 27001, GCP, Part 11/Annex 11, GDPR/HIPAA
Sponsors and inspectors care about resilience they can verify—not slideware. ISO 27001’s risk-based backbone translates into the operational discipline GxP expects. When ownership, cadence and evidence stay visible, responses land faster and third-party exposure narrows.
How ISO-First Maps to ICH E6(R2/R3) GCP
- Quality & oversight: Risks tie to controls across clinical ops, data mgmt, biometrics and PV; management reviews record decisions and CAPA.
- Study-level traceability: DoA logs, monitoring visit reports and follow-ups linked to systems and owners.
- TMF completeness: eTMF essentials and DIA TMF RM mapping with trends and exceptions.
How ISO-First Maps to Part 11 / Annex 11 / GAMP 5
- Validation packages: URS/FS/DS, test scripts, IQ/OQ/PQ, and traceability matrices in one place.
- Change control: Mid-study updates with approvals, diffs, rollback evidence and periodic reviews.
- Audit trails: Scheduled review workflows with sign-offs and exception handling.
How ISO-First Maps to GDPR/ISO 27701 & HIPAA
- Privacy records: RoPA, DPIAs, DSR logs tied to assets/services and study scope.
- Data transfers: DTA templates/logs; cross-border records and DPAs with vendors.
- Training & attestations: GxP, privacy and security in one lifecycle with reminders.
How ISO-First Maps to GVP / E2B(R3) & ISO 14155
- Safety case processing: ICSR/SUSAR evidence, vendor SLAs and audit trails.
- Device studies: Alignment with ISO 14155 GCP for medical devices.
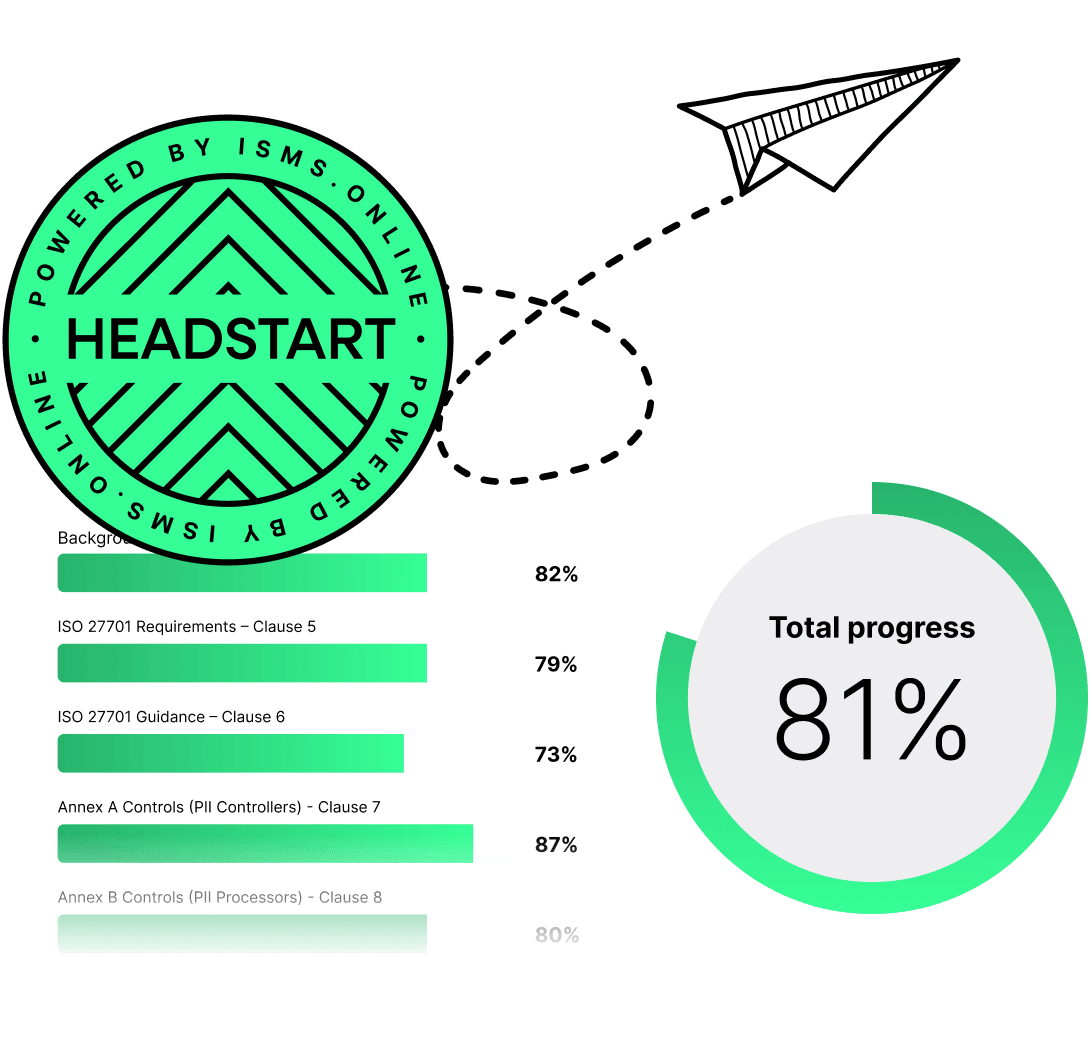
ISO 27001 made easy
An 81% Headstart from day one
We’ve done the hard work for you, giving you an 81% Headstart from the moment you log on. All you have to do is fill in the blanks.

Risk Management That Actually Runs for Contract Research Organisations (CROs)
Risk work should move every week, not just at inspection time. Linked risks, controls, assets and owners clarify accountability; consolidated views improve leadership decisions. Evidence reuse speeds inspections and sponsor audits, while management reviews drive continuous improvement without fire drills.
- Identify: Capture risks at org/program/study level; include RBQM signals (KRIs/QTLs); link to owners and controls.
- Treat: Assign actions, map to controls and CAPA, set due dates; maintain a traceable history that becomes ready-made evidence.
- Monitor: Run recurring checks—TMF completeness, audit-trail reviews, validation periodic reviews, training, privacy records, vendor SLAs—and collect artefacts for reuse.
- Review: Hold scheduled management reviews; record decisions, risk acceptances and exceptions to steer priorities.
- Report: Use trends (TMF %, query age, findings) to brief executives and sponsors.
- Renew: Roll forward linked evidence and SoA changes so inspections, renewals and sponsor assessments move faster.
An ISO-first operating system turns risk into a weekly workflow—ownership stays clear, evidence stays current, and decisions stay defensible.
A Features Checklist – What You Should Look For
CIO / IT Director
- ISO-first backbone prevents evidence sprawl and keeps one source of truth.
- Integrations act as data feeders; the ISMS governs cadence and ownership.
- Central RBAC/access and change history across EDC/eTMF/CTMS/IRT/LIMS/DCT.
Head of QA / CSV Lead
- Complete validation packages and traceability; Part 11/Annex 11 periodic reviews.
- CAPA and supplier audits mapped to systems and studies.
- Exportable inspection packs aligned to regulator and sponsor requests.
Head of Clinical Operations
- TMF completeness dashboards and monitoring packs.
- DoA and site oversight records with reminders and approvals.
- Study-level narrative ready for inspectors and sponsors.
Data Management Lead
- Audit-trail review workflow and sign-offs; reconciliation and DTA logs.
- Lock/closeout readiness views; exception handling and CAPA.
Biostats & Programming Lead
- Model/code change governance for SAP and outputs; CDISC SDTM/ADaM packages.
- Release diffs, approvals and rollback evidence.
Pharmacovigilance / Safety Lead
- ICSR/SUSAR case evidence; vendor SLAs and audit trails.
- Signal detection to CAPA traceability.
DPO / Privacy Lead
- RoPA/DSR/DPIA records with owners and audit trail.
- Cross-border transfers and DPAs maintained centrally.
- Policy lifecycle with versioning, approvals and attestations.
Capability Comparison for Contract Research Organisations (CROs)
| Capability | Why it Matters to CROs | What Good Looks Like |
|---|---|---|
| ISO-first system of record | Reduces evidence sprawl; one narrative for inspectors and sponsors | Repository linking risks, controls, assets, owners and evidence |
| Dynamic Statement of Applicability | Speeds inspector/sponsor Q&A | Live SoA with statuses, rationales and change history |
| Linked objects & RACI | Clarifies ownership and strengthens decisions | Bi-directional links; assignees; deadlines; traceable CAPA |
| Management reviews workspace | Sustains governance cadence and measurable improvement | Scheduled reviews with decisions, exceptions and actions |
| Evidence reuse & inspection packs | Accelerates inspections and sponsor audits | On-demand exports mapped to controls, periods and requests |
| Supplier/TPRM oversight (EDC/eCOA/LIMS/IRT) | Reduces third-party and data-integrity risk | Tiering, assessments, obligations and monitoring tied to services |
| Policy/SOP lifecycle & attestations | Prevents drift and inconsistent execution | Versioning, approvals, attestations, review reminders |
| Change control & validation (CSV/CSA) | Keeps mid-study changes compliant | Approvals, diffs, IQ/OQ/PQ evidence, periodic reviews |
| Audit-trail review & Part 11/Annex 11 | Demonstrates data-integrity oversight | Scheduled AT reviews with sign-offs and exceptions |
| TMF completeness & DIA TMF RM | Avoids closeout surprises | Completeness metrics, exceptions and exportable summaries |
| Privacy records (GDPR/HIPAA/27701) | Supports sponsor and DPA obligations | RoPA, DPIAs, DSR logs, DTAs and DPAs in one place |
| BCP/DR & scenario tests (22301) | Underpins operational resilience | Linked BIAs, test results, remediation and re-tests |
| RBQM (KRIs/QTLs) & trends | Focuses effort where risk rises | Thresholds, alerts and study-level roll-ups |
| Sponsor/regulator export packs | Cuts follow-ups and close-outs | Pre-mapped narratives and evidence bundles |

Free yourself from a mountain of spreadsheets
Embed, expand and scale your compliance, without the mess. IO gives you the resilience and confidence to grow securely.

Benefits You Can See in 90–180 Days
Shift from inspection sprints to a steady operating rhythm that compounds value across studies, audits and oversight.
- Faster inspection readiness (FDA/EMA/MHRA): Linked work shrinks requests and consolidates responses.
- Lower audit drag & cost: Continuous readiness removes last-minute scrambles.
- Stronger sponsor trust & faster awards: One narrative of control increases confidence.
- Predictable locks & closeouts: Stable cadence, TMF monitoring and reusable evidence.
- Team momentum: Clear owners, scheduled reviews and CAPA tracking keep improvements moving week by week.
- Framework reuse: The same risks, controls and evidence carry across GCP, Part 11/Annex 11, GDPR/HIPAA/27701, SOC 2, ISO 22301—without parallel paperwork.
- Cleaner validation & change governance: Approvals, diffs and periodic reviews lower findings.
When risks, controls and evidence live in one system of record, inspection packs assemble from the work itself and stakeholders can verify readiness at a glance.
Best ISO 27001 Compliance Software for CROs — A Quick Shortlist
ISMS.online ⭐
An ISO-first system of record designed to run the ISMS—not just pass an audit. Guided workflows link risks, assets, controls, owners and evidence so sponsor questionnaires shrink and reviews stay predictable.
A dynamic SoA, management reviews, and exportable inspection/sponsor packs keep readiness continuous across ISO 27001 today and GCP, Part 11, Annex 11, GAMP 5, GDPR, ISO 27701, HIPAA, SOC 2, ISO 22301 and ISO 14155 tomorrow. Connectors can feed artefacts; the ISMS keeps the governance cadence.
Vanta
Automation-forward with strong integrations and continuous tests that improve artefact collection speed. Great for gathering evidence quickly; you still define policy lifecycle, ownership and reviews to sustain ISO 27001 maturity.
Drata
Polished automation and monitoring with a broad connector story that accelerates collection. Helpful for evidence gathering; plan your management rhythm so governance and corrective actions don’t fall through the cracks.
Sprinto
Price-forward automation with a wide integration surface that moves quickly from zero to audit. A pragmatic on-ramp; long-term outcomes rely on clear owners, milestones and recurring management reviews.
Secureframe
Automation plus questionnaires and trust-centre features at higher tiers can speed diligence. Ensure your internal cadence—reviews, internal audits and CAPA—remains the backbone of maturity.
DataGuard
Hybrid software + services model is useful when internal capacity is thin. Weigh commercial complexity and keep one authoritative system of record for day-to-day operation.
Strike Graph
Automation/GRC-lite proposition with public pricing offers a solid entry point. Validate how risks, controls and evidence roll up into a management-ready narrative stakeholders will trust.
HiComply
Template-led approach with transparent tiers speeds initial drafting. Lasting value comes from clear ownership, traceability, and a steady review cadence across the year.
See the ISMS.online Platform in Action
A live ISMS.online walkthrough shows end-to-end traceability across risks, controls, owners and evidence.
You’ll see how a linked Statement of Applicability speeds inspector and sponsor responses, how a steady governance rhythm sustains improvement, and how cross-mapped evidence helps you reuse work across GCP, Part 11/Annex 11, GDPR/27701 & HIPAA, SOC 2, ISO 22301, ISO 14155 without duplicate projects.
Find out more by booking a demo today.
Frequently Asked Questions
What makes compliance software “CRO-ready”?
An ISO-first backbone that links risks, controls, owners and evidence; live SoA; validation & change control; audit-trail review; TMF completeness; privacy records; RBQM; and exportable inspection/sponsor packs.
How fast can we see value?
Most teams establish cadence within 90–180 days when owners, reviews and CAPA are scheduled from day one. Linked work shortens questionnaires and lowers audit effort.
What should we see on a demo to confirm traceability?
A live ISMS overview linking a risk → control → owner → current evidence, the corresponding SoA entry and rationale, plus an exportable inspection pack aligned to GCP and Part 11/Annex 11.
How does this map to GCP, Part 11/Annex 11, GDPR/27701 & HIPAA?
Risk-based controls align to quality, validation and privacy themes; review schedules support governance obligations; cross-mapped evidence lowers the time to add frameworks without duplicate projects.
How do we handle audit-trail reviews and validation periodic reviews?
Schedule review workflows with sign-offs and exception handling; keep IQ/OQ/PQ artefacts and diffs linked to changes and studies.
What about DCT vendors (eCOA/ePRO/eConsent) and data transfers?
Use supplier tiering and DTA/transfer logs with obligations, SLAs and monitoring tied to services. Link findings to CAPA.
Can we reuse effort across ISO 27001, GCP, Part 11/Annex 11, GDPR/HIPAA, SOC 2 and ISO 22301?
Yes. One narrative of control with mapped requirements lets evidence and owners serve multiple frameworks—without parallel paperwork.
What are typical cost drivers?
Seats, frameworks in scope, assurance depth (evidence history, SoA detail, supplier oversight), number of programmes/studies and integrations.
What does implementation look like?
Scope services and assets (EDC, eTMF, CTMS, IRT, LIMS, DCT, analytics), import policies and risks, link controls and evidence, set your review calendar and assemble inspection packs directly from the work.
Does this replace our eQMS/GRC or ticketing tools?
Keep eQMS/ticketing for quality and work management. Use integrations as feeders; let the ISMS hold the authoritative story of risks, controls, evidence and ownership.
How do we prepare for the next inspection or sponsor audit?
Continuous reviews, internal audits and corrective actions build re-usable inspection packs. Predictable cadence stabilises effort and timelines year over year.









